GMP Certification is a term that is recognized worldwide for the control and management of manufacturing, testing and overall quality control of food and pharmaceutical products. GMP takes quality assurance approach, which ensures that products are consistently produced and controlled to the quality standards appropriate to their intended use and as required by the marketing authorization.
Good manufacturing practice” or “GMP” is part of a quality system covering the manufacture and testing of active pharmaceutical ingredients, diagnostics, foods, pharmaceutical products, and medical devices. GMPs are guidelines that outline the aspects of production and testing that can impact the quality of a product. Many countries have legislated that pharmaceutical and medical device companies must follow GMP procedures and have created their own GMP guidelines that correspond with their legislation.
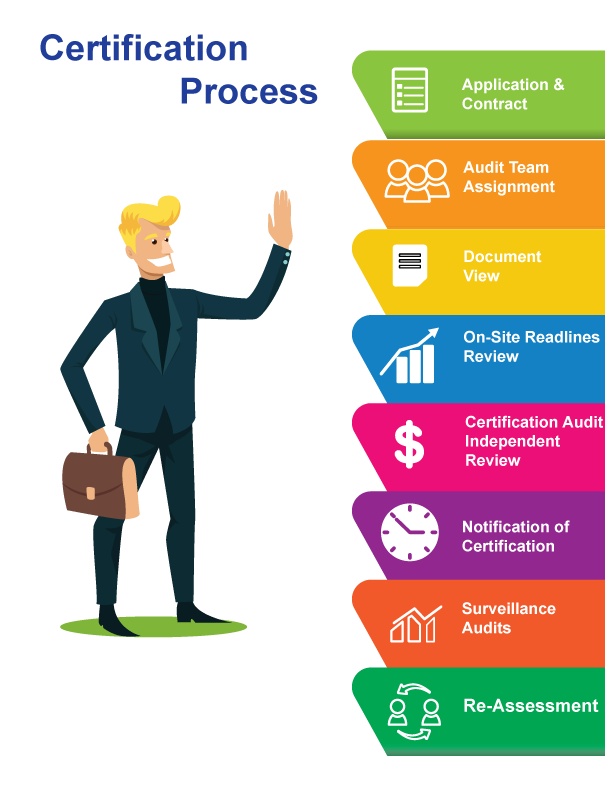
ACHIEVING GMP CERTIFICATION
- We can also provide a no-obligation competitive quotation from our dedicated business development team.
- Our leading auditors will devise a comprehensive assessment program which will be agreed in line with your GMP requirements.
GMP Resources
Good Manufacturing Practice (GMP) is a system for ensuring that products are consistently produced and controlled according to quality standards. It is designed to minimize the risks involved in any pharmaceutical production that cannot be eliminated through testing the final product.
GMP covers all aspects of production from the starting materials, premises, and equipment to the training and personal hygiene of staff. Detailed, written procedures are essential for each process that could affect the quality of the finished product. There must be systems to provide documented proof that correct procedures are consistently followed at each step in the manufacturing process – every time a product is made.
CERTIFICATION BENEFITS
-
- Eligibility for submitting tenders or proposals for projects.
- Generates new business opportunities
- Can boost your organisation’s brand reputation and be a useful promotional tool, especially when going up against competitors who aren’t certified
- Attract investment and improve brand reputation
- Improve corporate image. Project international quality in your services.
- Increased customer confidence and satisfaction
- Strengthen effectiveness and efficiency
- Enhance internal effectiveness and improve productivity performance through elimination of unnecessary costs
- Minimize corporate risk



